The Vital Role of Phlebotomists in Blood Testing: Regulations, Guidelines, and Standing Orders in the United States
Summary
- Standing orders for common tests are allowed in the United States, providing convenience to patients and Healthcare Providers.
- Phlebotomists play a crucial role in performing blood tests and ensuring accuracy in the results.
- Regulations and guidelines are in place to ensure the safety and quality of Blood Testing procedures.
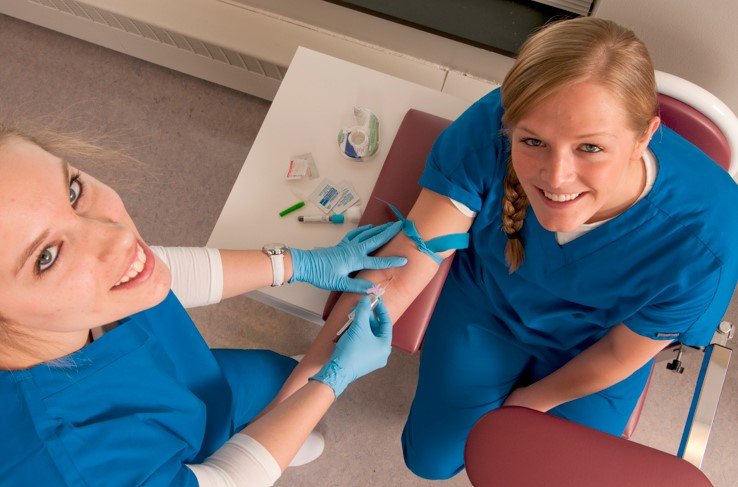
The Role of Phlebotomists in Blood Testing
Phlebotomists are healthcare professionals responsible for drawing blood from patients for various medical tests and procedures. They play a crucial role in the healthcare system by ensuring that blood samples are collected accurately and safely. Phlebotomists work in various healthcare settings, including hospitals, clinics, laboratories, and blood donation centers.
Training and Certification
Phlebotomists undergo specialized training to learn proper techniques for Venipuncture, the process of drawing blood from a vein. They are also trained to handle blood samples, label them correctly, and ensure that they are transported safely to the laboratory for testing.
- Phlebotomy training programs are available at vocational schools, community colleges, and technical institutes.
- After completing their training, phlebotomists may choose to become certified through organizations such as the American Society for Clinical Pathology (ASCP) or the National Healthcareer Association (NHA).
- Certification demonstrates a phlebotomist's commitment to their profession and ensures that they have met specific competency standards.
Importance of Accuracy in Blood Testing
Accuracy in Blood Testing is essential for making accurate diagnoses, monitoring patients' health conditions, and guiding treatment decisions. Phlebotomists play a critical role in ensuring that blood samples are collected correctly to prevent errors in Test Results.
- Proper Venipuncture techniques help minimize the risk of sample contamination or hemolysis, which can affect the accuracy of Test Results.
- Phlebotomists must follow strict protocols for labeling blood samples and documenting the time and date of collection to ensure traceability and prevent mix-ups.
- Training and certification help phlebotomists develop the skills and knowledge needed to perform blood draws accurately and efficiently.
Regulations and Guidelines for Blood Testing
In the United States, Blood Testing is regulated by various federal and state agencies to ensure the safety and quality of laboratory procedures. These Regulations govern the collection, handling, and analysis of blood samples to protect patients and Healthcare Providers from potential risks.
CLIA Regulations
The Clinical Laboratory Improvement Amendments (CLIA) regulate all laboratory testing performed on humans in the United States. CLIA standards apply to all clinical laboratories, including those that perform Blood Testing for diagnostic purposes.
- CLIA Regulations establish Quality Control measures to ensure the accuracy and reliability of Test Results.
- Laboratories must meet specific Proficiency Testing requirements to demonstrate their ability to perform accurate testing.
- Inspections are conducted regularly to assess laboratories' compliance with CLIA standards and identify any deficiencies that need to be addressed.
Professional Guidelines
Professional organizations, such as the American Society for Clinical Laboratory Science (ASCLS) and the American Medical Technologists (AMT), provide guidelines and best practices for phlebotomists and other healthcare professionals involved in Blood Testing.
- Guidelines cover topics such as infection control, specimen collection procedures, and patient safety to ensure that Blood Testing is conducted in a safe and efficient manner.
- Phlebotomists are encouraged to stay up-to-date with the latest guidelines and recommendations to maintain the highest standards of practice.
- Continuing Education opportunities are available to help phlebotomists enhance their skills and knowledge in Blood Testing and specimen collection.
Standing Orders for Common Tests
Standing orders for common tests allow Healthcare Providers to order specific laboratory tests for patients without the need for individual consultations or prescriptions. This practice helps streamline the testing process and improve access to essential healthcare services for patients.
Benefits of Standing Orders
Standing orders for common tests offer several benefits for patients, Healthcare Providers, and laboratory staff:
- Convenience: Patients can receive necessary tests without the need for multiple appointments or consultations, saving time and resources.
- Timely treatment: Standing orders help Healthcare Providers make quick decisions about patients' care based on the results of essential laboratory tests.
- Efficiency: Laboratory staff can process samples quickly and prioritize urgent tests when standing orders are in place for common tests.
Legal and Ethical Considerations
While standing orders for common tests are allowed in the United States, Healthcare Providers must follow legal and ethical guidelines to ensure patient safety and confidentiality:
- Authorized practitioners: Only licensed Healthcare Providers can order laboratory tests under standing orders, ensuring that tests are ordered appropriately and for VALID medical reasons.
- Patient Consent: Healthcare Providers must obtain Informed Consent from patients before ordering tests under standing orders to ensure that patients understand the purpose of the tests and their potential implications.
- Confidentiality: Laboratory staff must adhere to strict confidentiality protocols to protect patients' privacy and ensure that Test Results are shared only with authorized individuals.
Quality Assurance and Monitoring
To maintain the quality and integrity of laboratory testing under standing orders, Healthcare Providers and laboratory staff must implement quality assurance measures and monitor the process regularly:
- Documentation: Healthcare Providers must document the reasons for ordering tests under standing orders and the results of the tests in patients' medical records for continuity of care.
- Review process: Healthcare organizations should establish a review process to evaluate the appropriateness of standing orders for common tests and make adjustments as needed to ensure patient safety and quality of care.
- Feedback mechanism: Patients should have the opportunity to provide feedback on their testing experience under standing orders to help Healthcare Providers improve services and address any concerns.
Conclusion
Standing orders for common tests play a vital role in providing convenient access to essential healthcare services for patients in the United States. Phlebotomists contribute to the accuracy and safety of Blood Testing procedures, ensuring that patients receive reliable Test Results for diagnosis and treatment. By following Regulations, guidelines, and best practices, Healthcare Providers can maintain the quality and integrity of laboratory testing under standing orders, ultimately improving patient care and outcomes.
Disclaimer: The content provided on this blog is for informational purposes only, reflecting the personal opinions and insights of the author(s) on the topics. The information provided should not be used for diagnosing or treating a health problem or disease, and those seeking personal medical advice should consult with a licensed physician. Always seek the advice of your doctor or other qualified health provider regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website. If you think you may have a medical emergency, call 911 or go to the nearest emergency room immediately. No physician-patient relationship is created by this web site or its use. No contributors to this web site make any representations, express or implied, with respect to the information provided herein or to its use. While we strive to share accurate and up-to-date information, we cannot guarantee the completeness, reliability, or accuracy of the content. The blog may also include links to external websites and resources for the convenience of our readers. Please note that linking to other sites does not imply endorsement of their content, practices, or services by us. Readers should use their discretion and judgment while exploring any external links and resources mentioned on this blog.
